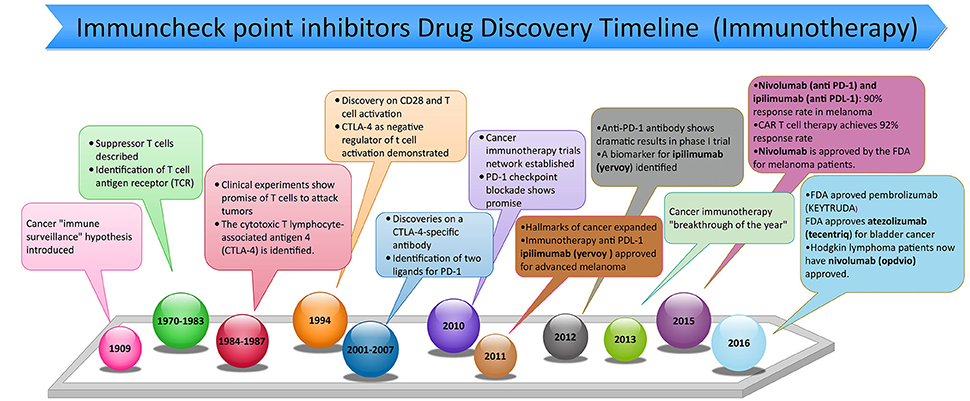
Frontiers | PD-1 and PD-L1 Checkpoint Signaling Inhibition for Cancer Immunotherapy: Mechanism, Combinations, and Clinical Outcome

Study Comparing the Combination Domvanalimab and Zimberelimab With Pembrolizumab in Untreated Locally Advanced or Metastatic PD-L1-High Non-Small Cell Lung Cancer
Frontiers | Neuro-ophthalmic complications of immune checkpoint inhibitor therapy: Current status and future directions
Merck receives EC approval for Keytruda to treat relapsed Hodgkin Lymphoma - Pharmaceutical Technology
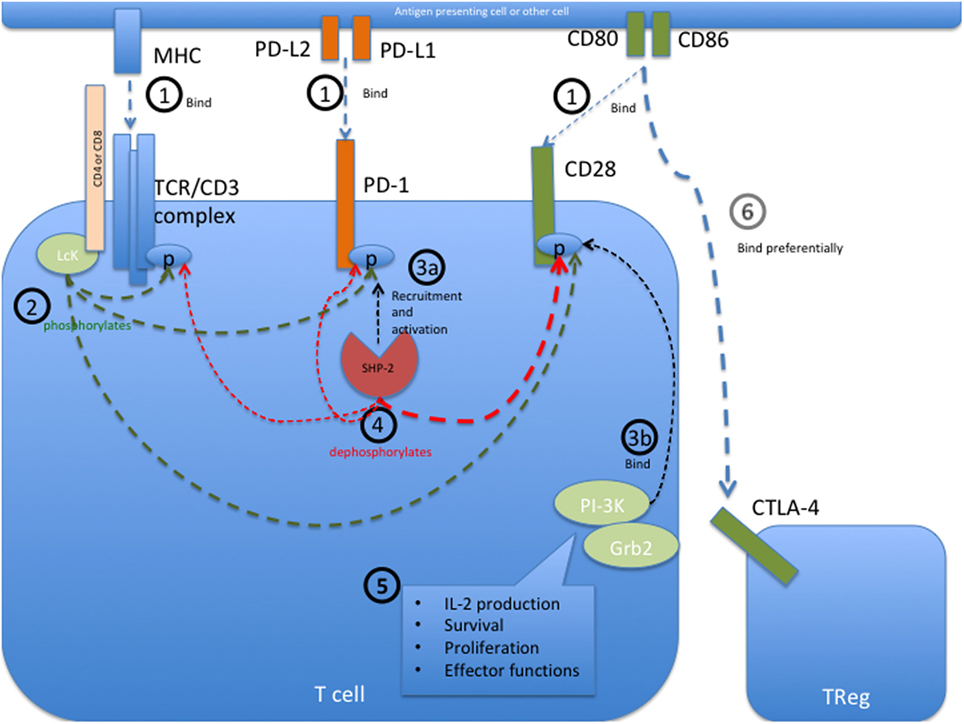
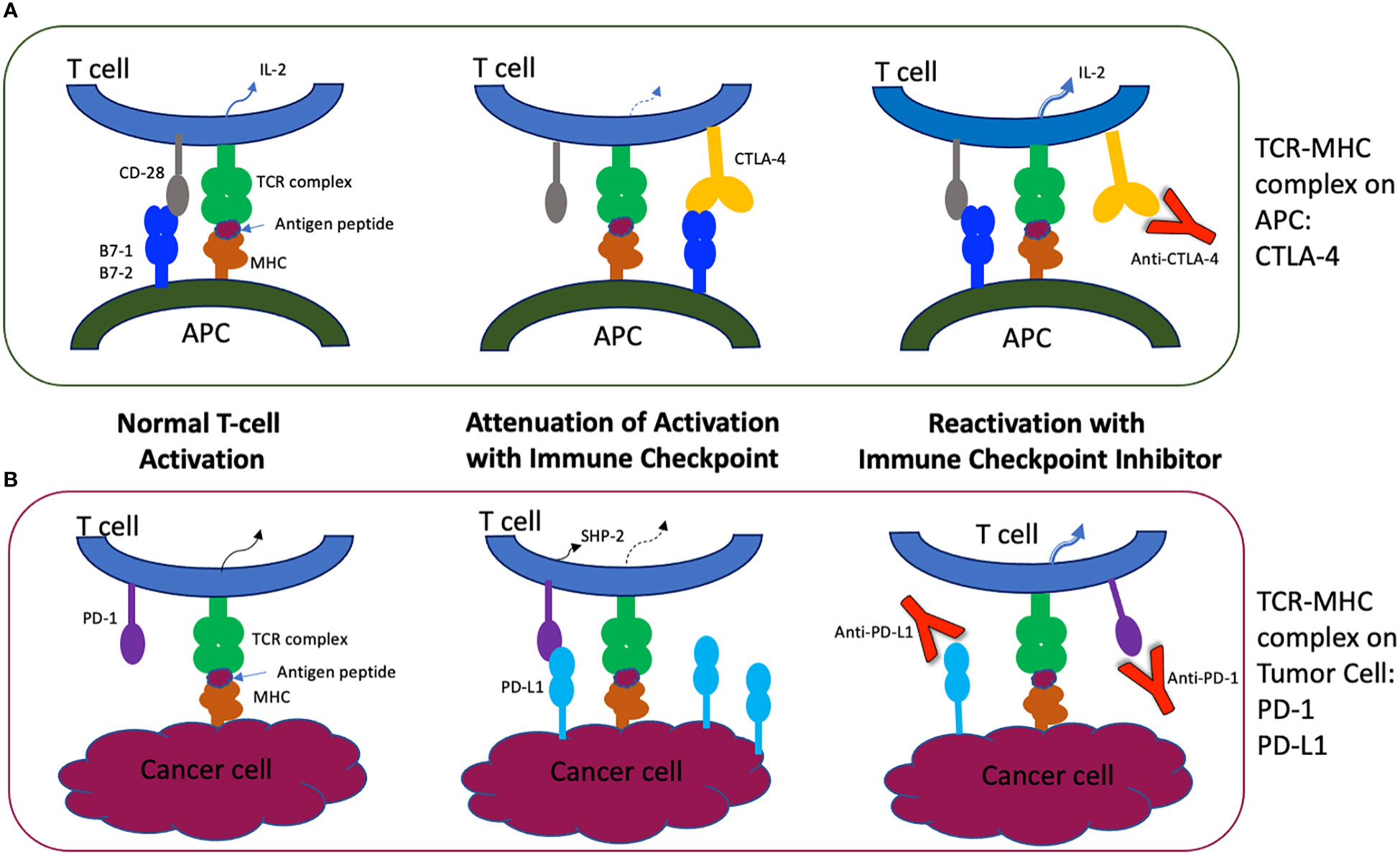
Frontiers | Anti-PD-1 and Anti-CTLA-4 Therapies in Cancer: Mechanisms of Action, Efficacy, and Limitations

FDA Approves Merck's KEYTRUDA® (Pembrolizumab) for First-Line Treatment of Patients with Unresectable or Metastatic MSI-H or dMMR Colorectal Cancer, First Single-Agent, Anti-PD-1 Therapy Approved for the First-Line Treatment of These Patients -

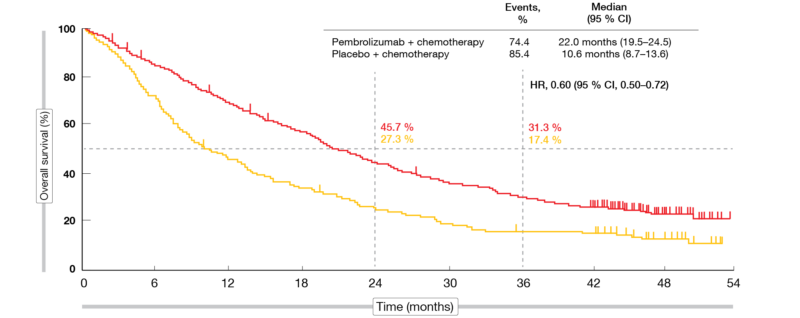
Updated Analysis of KEYNOTE-024: Pembrolizumab Versus Platinum-Based Chemotherapy for Advanced Non–Small-Cell Lung Cancer With PD-L1 Tumor Proportion Score of 50% or Greater | Journal of Clinical Oncology

Second-line therapy with nivolumab plus ipilimumab for older patients with oesophageal squamous cell cancer (RAMONA): a multicentre, open-label phase 2 trial - The Lancet Healthy Longevity
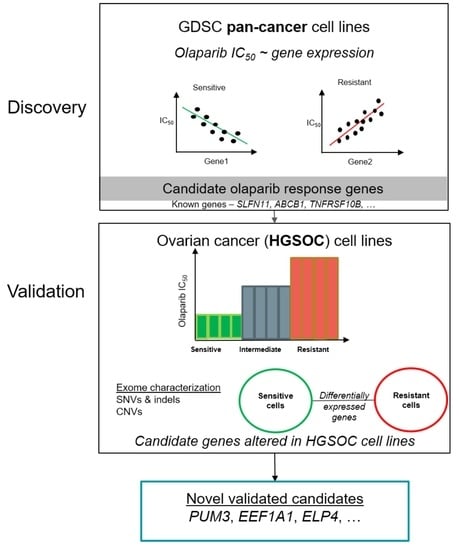
Cancers | Free Full-Text | Candidate Markers of Olaparib Response from Genomic Data Analyses of Human Cancer Cell Lines